For Research Use Only. Not for human or veterinary use.
Lactacystin (CAS: 133343-34-7)Product ID: BLK-0460 CAS 133343-34-7 Mechanism Primary Target Pathway | |
| Structure |  |
| CAS | 133343-34-7 |
| Molecular Formula | C15H24N2O7S |
| Molecular Weight | 376.43 |
| Price | 1mg: USD 320.00 / 5mg: - / 25mg: - |
| Storage | -20°C |
| Solubility | 1mg/ml ethanol, methanol, DMSO |
| Synonyms | Clasto-lactacystin β-lactone |
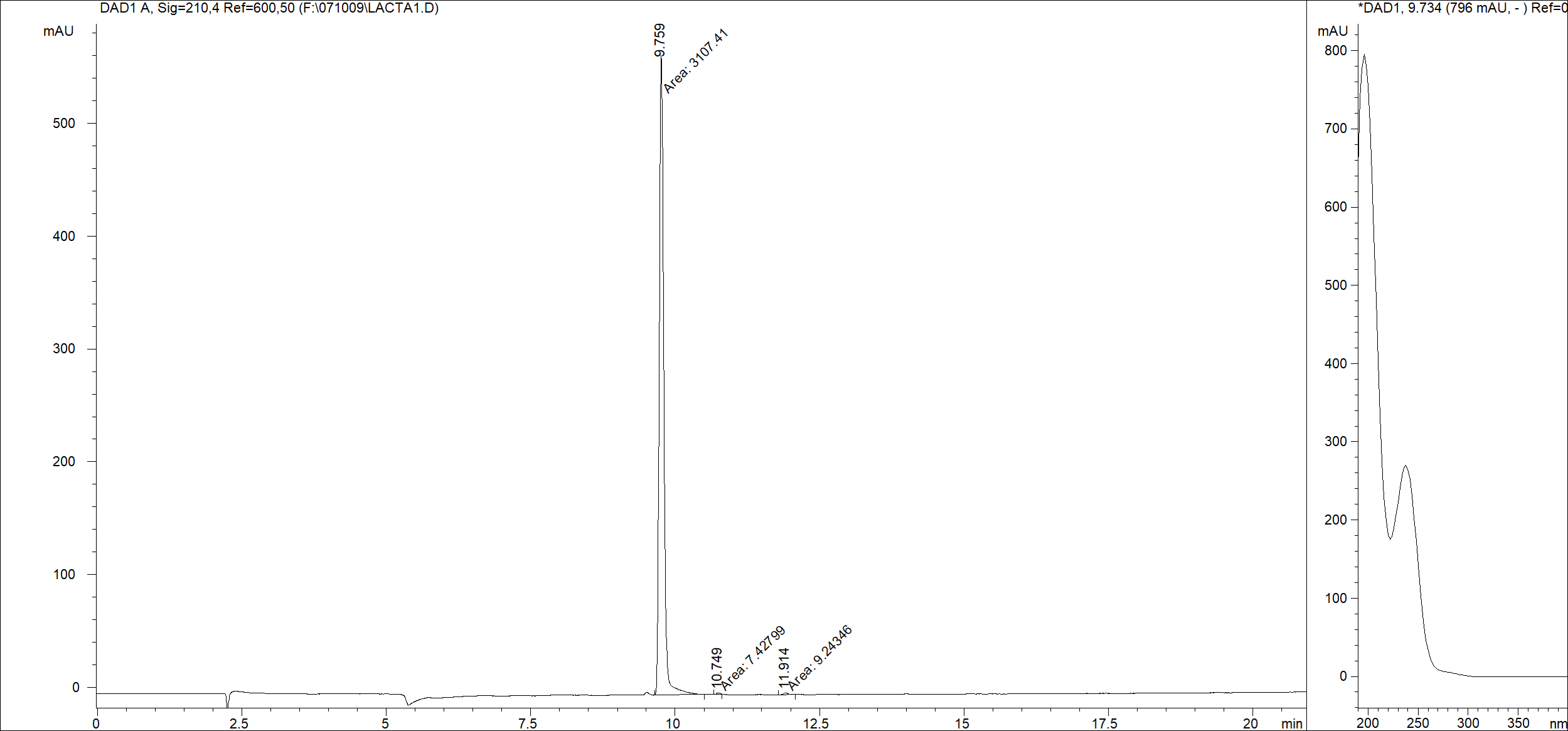
| Purity | >95% [View LC/UV Data] |
| LC/UV | Representative LC/UV chromatogram under typical analytical conditions (C18 column; mobile phase: 0–40% ACN / H2O (0.05% H3PO4), linear gradient 20 min; flow rate: 0.2 mL/min; detection: 210 nm). |
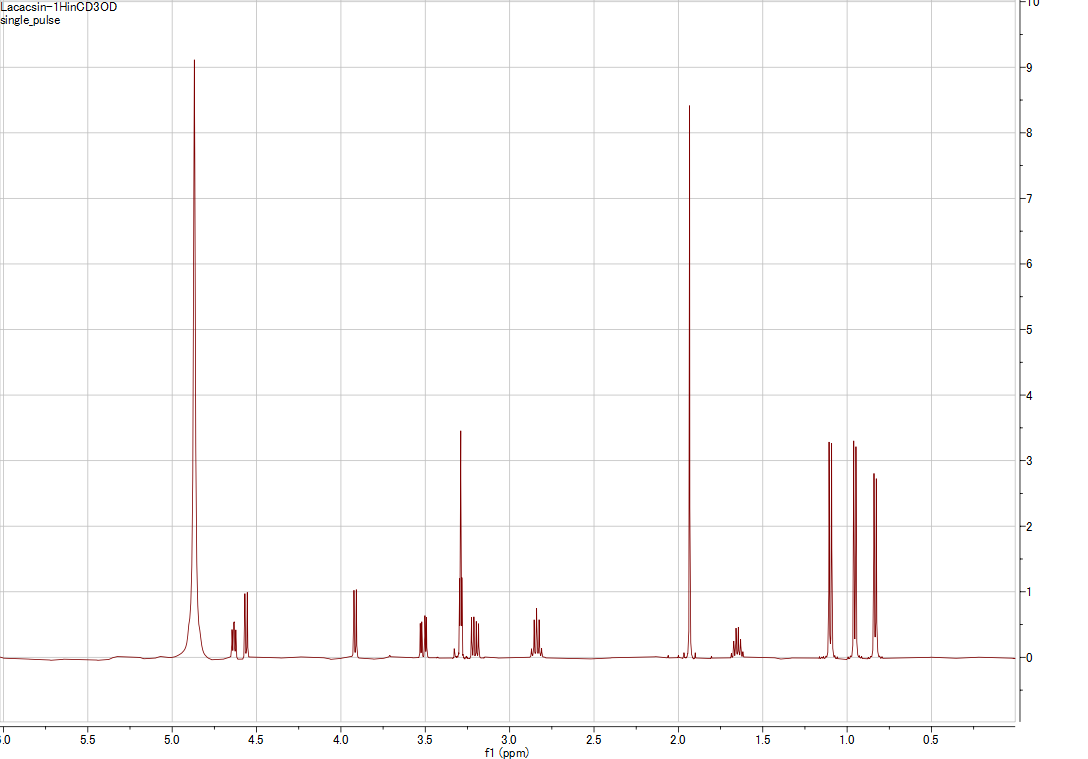
| NMR Data | 1H NMR (CDCl3, 500 MHz) [View] [Full page] |
| NMR Spectral Data | Representative 1H NMR (CDCl3, 500 MHz) spectrum confirming the structure of Lactacystin. Representative 13C NMR (CDCl3, 125 MHz) spectrum supporting structural assignment of Lactacystin. |
| Source Organism | Streptomyces lactacystinaeus. |
| Summary | Lactacystin is a Streptomyces-derived natural product that irreversibly inhibits the proteasome. |
| Details | |
| Browse Related | Mechanism Primary Target Chemical Class |
| Mechanism | Proteasome inhibitor |
| Primary Target | 20S proteasome (? subunits) |
| Pathway | Ubiquitin–proteasome pathway |
| Chemical Class | ?-Lactone |
| Discovery Year | 1991 |
| Keywords | |
| References |
|
Request pricing, availability, or bulk supply information for this compound. Get a QuoteGeneral Inquiry | |
Documents | |
| ← Back to Catalog | |